Making Liquid Fuels From Natural Gas: A Technological Challenge Of The Twenty First Century
Ai-Quoc Pham
Lawrence Livermore National Laboratory
Chemistry & Materials Science
P.O. Box 808, L-231
Livermore, CA 94550
Tel: (510)-423-3394
e-mail: [email protected]
Abstract
Research on alternative feedstock utilization is one of the key technologies of the twenty first century because the world petroleum reserve is anticipated to run out within the next fifty years. Natural gas, for which the reserve is almost as abundant as for fossil oils, is one of the potential substitutes that is currently under-exploited. This talk will give a brief overview of current natural gas exploitation technologies, then will discuss a new technology currently developed by Lawrence Livermore National Laboratory and others. This new technology is based on dense ceramic electrochemical membranes that selectively permeates oxygen from air to the natural gas side where the selective oxidation occurs. The resulting mixture of carbon monoxide and hydrogen gases, called syn-gas, is a precursor that can be used to synthesize valuable chemicals such as methanol and liquid fuels for transportation.
Energy Consideration
Although considerable efforts have been devoted to the development and exploitation of alternative energy, the world economy is still heavily dependent on petroleum supply. The share of petroleum in the total energy consumption is still 40% in 1995, down by only 8% from the energy crisis of 1973. The absolute petroleum consumption has actually increased by 50% during the same time period. Considering the current energy consumption growth, studies have shown that the world petroleum reserve will be depleted within the next century. Research on alternative feedstock utilization is thus one of the key technologies of the twenty first century for the survival of the world.
Although nuclear energy seems to be an abundant and inexhaustible energy source, its actual share to the total energy production is anticipated to decrease in the next 20 years. The origin of this decrease is attributed to the numerous problems associated with the fission reaction, especially the waste management that has never had an environmentally acceptable solution.
Actually, because of many similar properties with petroleum, which makes easy the use of existing infrastructure, natural gas appears to be the most promising alternative candidate. The world’s total natural gas reserve is estimated to be more than 5,000 TCF (trillion cubic feet), half of which is in remote region. In energy unit, the world proved reserves of natural gas is about 4127 EJ (ExaJoules, 1 EJ = 1018 J ) almost equal to the amount of 4749 EJ for crude oil.
Current uses of natural gas
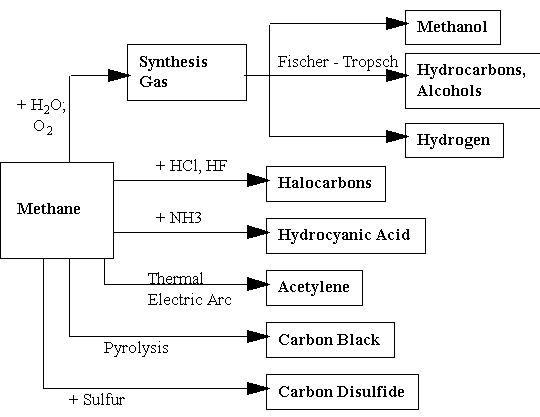
In 1994, the U.S. gas consumption totaled 20.8 TCF, mainly for domestic and industrial heating. The low energy content per unit volume of natural gas makes its transport from wellheads to the consumers difficult, limiting the widespread use of natural gas. Because of the low energy efficiency of the combustion process, about 30% in general, heating is an under-use of natural gas. It is much more desirable to upgrade natural gas to other chemicals that have important applications in petrochemical industry or in transportation. One can distinguish six reactions of current industrial importance for the direct conversion of methane which is the principal constituent of natural gas (figure 1):

Figure 1: Uses of natural gas (methane) in industrial processes.
The most important one is the conversion of natural gas to synthesis-gas, a mixture of CO and H2. This gas is an extremely important precursors for the synthesis of methanol, hydrogen, ammonia and other products. In particular, methane can be converted to synthesis gas and gasoline can then be produced from this mixture by the Fischer-Tropsch (FT) process. Gasoline is currently produced in Sasol, South Africa and in Malaysia using this technology. Methanol can be produced from synthesis gas using the so called MTG process of Mobil.
Currently, only about 7% of the world natural gas production is used in chemical industries. Only 1.4% is used to produce liquid fuel. The reason why so little natural gas is converted to liquid fuel is the high cost of existing conversion technologies that makes the gasoline produced by either process much more expensive than gasoline obtained from crude oil. Conventional methods are very energy and capital intensive, because they operate at high pressures and temperatures. Another method, namely the partial oxidation using pure oxygen, is also expensive because a costly cryogenic oxygen plant is required. The synthesis gas produced using existing methods accounts for about 60% of the total conversion cost of natural gas to gasoline.
Thus, natural gas is an excellent candidate as an abundant alternative energy source. To make possible the wide spread use of natural gas, the conversion cost must be lowered. We are working on the development of a new technology that can potentially reduced by 50% the cost of synthesis gas production. This new technology is based on dense ceramic membranes that can selectively permeate oxygen. The membrane would be used to separate air from the natural gas side. Oxygen permeated through the membrane will undergo a partial oxidation reaction with natural gas to yield CO and H2, the synthesis gas.
Ceramic membranes
The most common form of electrical conduction is via electrons. However, some materials can also exhibit ionic conduction under certain conditions. For example, the oxygen ion conduction in certain oxide materials such as zirconia have been known since Nernst’s discovery in 1899. This property has been exploited in numerous applications such as the oxygen sensor which is currently used in the car for air/fuel ratio control or in fuel cells for clean electricity generation. In 1987, Teroaka discovered a new class of materials that exhibit both electronic and ionic conductions. The oxygen ion conduction in the new material, a perovskite phase of composition La1-xSrxFe1-yCoyO3, can be as high as 1 S.cm-1 at 800°C, about two order of magnitude higher than the ionic conduction of doped-zirconia. The high ionic and electronic conductions in this material make possible its use as an oxygen permeable membrane. When an oxygen concentration exists across the membrane, oxygen from the oxygen rich side will be reduced electrochemically to oxygen ion which is then transported through the ceramic membrane. Arriving on the oxygen lean side, oxygen ion will be reoxidized to oxygen molecule, which is liberated to the gas ambient. The electrons involved in the oxidation/reduction process is transported back to the other side, insuring the overall electroneutrality. Because the membrane is an oxygen ion conductor, only oxygen can be transported through the membrane, thus the selectivity of the permeation process is almost 100%. The principle of the membrane is illustrated in figure 2.
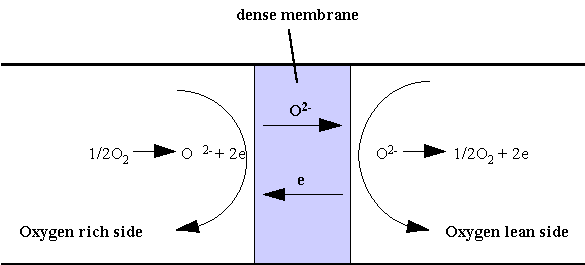
Figure 2: Principle of dense ceramic membrane.
Currently, ceramic membranes exhibit a maximum flux of a few milliliters of oxygen gas (in normal conditions) per square centimeter per minute. At Lawrence Livermore National Laboratory, we are developing the theoretical framework of the oxygen permeation process. Such a global description of the oxygen permeation through dense ceramic membrane was not available until now. The newly developed theory predicts that oxygen flux as high as 100 ml.cm-2.min-1 is possible if the surface of the membrane is coated with a high surface area catalyst layer (A > 50m2/g). Thus we are concentrated our effort on the development of this multi-layer type membrane (dense membrane with porous catalyst coating).
The oxygen permeable membrane can potentially substitute to the expensive cryogenic oxygen plants for oxygen production. When combined with an appropriate catalyst on the oxygen lean side, the membrane can be used to convert natural gas to synthesis gas via a partial oxidation reaction:
CH4 + 1/2O2 Þ CO + 2H2
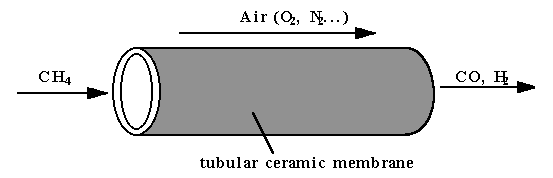
Figure 3: Schematic of a tubular ceramic membrane.
Figure 3 illustrates the principle of the conversion of natural gas to synthesis gas using tubular ceramic membrane. The tubular membrane is made by extrusion. Air is flowing outside of the tube while natural gas is flowing inside. The inside of the tube is coated with a Ni-based partial oxidation catalyst. The whole system is heated in a tubular furnace. The natural gas outlet side is monitored using a gas chromatography to analyze the composition. Figure 4 shows our preliminary results. Carbon monoxide selectivity higher than 95% was obtained in the temperature range of 700 to 820°C. The conversion was close to theoretical value deduced using thermodynamic data. Although we could achieve only 44% conversion in the present case, higher values, higher than 90% are possible. These preliminary results are extremely interesting and more experiments will be pursued in this direction.
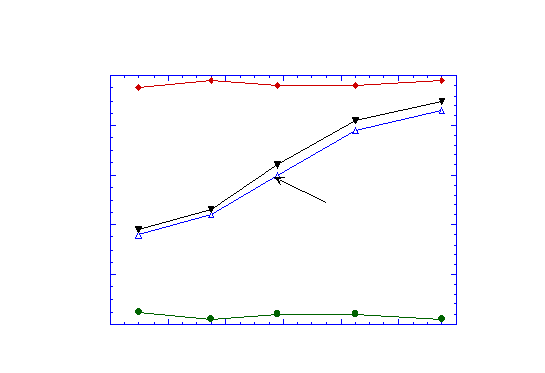
Figure 4: Conversion of methane to synthesis gas. The conversion factor is defined as the ratio of the number of moles of methane converted to the number of moles of methane introduced. The CO selectivity factor is the ratio of the number of moles of CO formed to the number of moles of methane introduced.
Conclusion
The shortage of petroleum is a clear certitude for the next century. Alternative energy sources are available but the exploitation technologies are in general not as mature as for petroleum. This has been shown to be the case of natural gas. Indeed, the feasibility of the upgrading of natural gas to valuable chemicals, especially liquid fuels has been known for years. However, the high cost of the steam reforming and the partial oxidation processes, used for the conversion of natural gas to synthesis gas, has hampered the widespread exploitation of natural gas.
Dense ceramic membranes were shown to be an exciting new and low cost technology for the upgrading of natural gas. This new technology will increase the utilization of domestic natural gas reserves through the conversion to liquid fuels and will decrease the nation’s dependence on imported oil. In longer term, natural gas can be substituted to petroleum.
References
1. International Energy Outlook: World Energy Outlook 1997.
2. C.N. Satterfield, "Heterogeneous Catalysis in Practice", McGraw-Hill chemical enginerring series.
3. Y. Teraoka, H. Zhang, S. Furukawa and N. Yamazoe, Chem. Lett. 1743 (1985).